Expected survival of Primary procedure for efficacy assessment is a non-surgical interventional procedure (e.g, lumbar puncture, skin biopsy, cardiac catheterization, endoscopic retrograde cholangio-pancreatography).Any surgical procedure that involves the intraoperative use of systemic, intravascular, unfractionated heparin.The patient has acute bleeding in a critical area or organ, such as pericardial, intracranial, or intraspinal.The patient has overt bleeding associated with a fall in hemoglobin level by ≥2g/dL, OR, a hemoglobin ≤8 g/dL if no baseline hemoglobin is available.The patient has acute-overt bleeding that is potentially life-threatening, e.g., with signs or symptoms of hemodynamic compromise, such as severe hypotension, poor skin perfusion, mental confusion, low urine output that cannot be otherwise explained.Surgery for which the risk of clinically meaningful uncontrolled or unmanageable bleeding is low.Īcute life-threatening bleeding (ISTH criteria) at the time of Screening:.If a patient meets any of the following criteria, he or she is not eligible: Willingness to use highly effective methods of contraception through 30 days following study drug dose (for female and male patients who are fertile). 
Have a negative pregnancy test documented prior to enrollment (for women of childbearing potential). 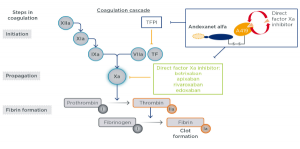
Note: Patients enrolled in this manner should receive a high-andexanet dosing regimen.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
